Editor’s note: After moving to weekly updates this spring, we’re restarting daily COVID-19 updates as the state experiences increased cases.
What you need to know this week: The U.S. Food and Drug Administration (FDA) gave its full approval of Pfizer’s COVID-19 vaccine Monday in individuals age 16 and up. Pfizer’s vaccine is the first to receive approval.
“The FDA’s approval of this vaccine is a milestone as we continue to battle the COVID-19 pandemic,” said acting FDA commissioner Dr. Janet Woodcock. “While millions of people have already safely received COVID-19 vaccines, we recognize that for some, the FDA approval of a vaccine may now instill additional confidence to get vaccinated.”
Law professor Lindsay Wiley from American University in Washington, D.C., told NPR it’s an open question right now as to whether having the full approval makes a difference in terms of legal authority to issue a vaccination requirement. But, experts expect a greater number of vaccine requirements put in place in schools and workspaces with FDA approval.
Until the FDA’s decision, the vaccine was able to be used under an emergency use authorization. Pfizer applied for full approval in May. The FDA still does not recommend that children under age 12 get the Pfizer vaccine.
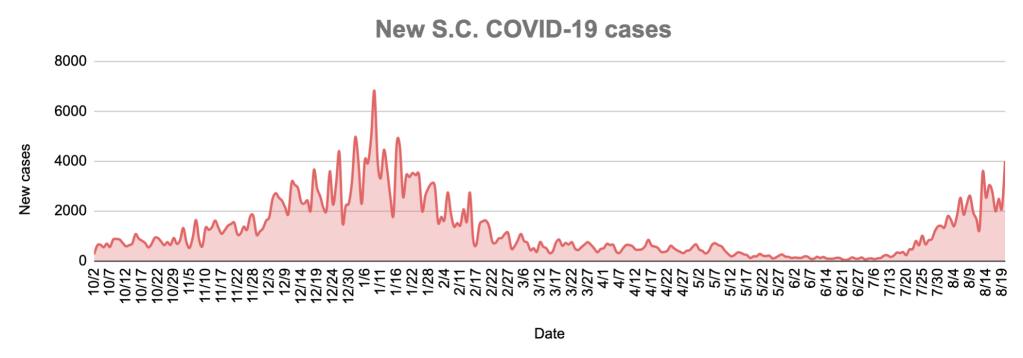
Latest COVID-19 data
South Carolina health officials reported 3,648 total cases of COVID-19 Aug. 24, with 3,121 confirmed. A total of 10 new deaths, with 9 confirmed, were reported Tuesday.
With 25,168 tests reported, 16.2% were confirmed positive.
Percentage of S.C. residents with at least 1 vaccine: 55.4%
Percentage of S.C. residents who have completed vaccination: 46.7%















